Artificial Intelligence for Pharma and Healthcare Services
How to transform the manual and fragmented management of GMP compliance into an automated, secure, and efficient process
The Problem
An analysis of over 180 Italian pharmaceutical companies shows that 72% of Quality Managers spend more than 12 hours/week on manual documentation management and audit preparation, resulting in inefficiencies that directly affect productivity and regulatory compliance (ISTAT, Industrial Production March 2025).
The increasing digitalization of pharma and the rise in regulations (FMD directive, EU serialization, GDP) require a systematic approach to address the following issues:
- Fragmented GMP document management: searching for technical and regulatory information takes 3-4 hours per week per operator, with a 15% risk of manual errors that can cause non-compliance during AIFA/EMA inspections.
- Complex audit preparation: collecting documentation for inspections requires 10-15 days of intensive work by dedicated teams, taking up to 25% of the time from main quality control processes.
- LIS-SAP-eCRF information silos: the presence of non-integrated systems generates 30% inefficiency in data flows, manual rework, and possible gaps in GDP traceability.
- Risk of using general-purpose AI: tools like ChatGPT or Copilot, not specialized in pharma, provide generic answers that can compromise the safety and compliance of GMP procedures.

The Solution
We have implemented an integrated Intelligence platform that transforms the pharmaceutical ecosystem into a compliant data-driven environment, where every process becomes a lever for traceable and verifiable optimization.
The integrated technology stack we have developed:
- GMP Compliance Engine: complete automation of document management with FDA/EMA-compliant audit trails, reducing audit preparation times by 60% and ensuring automatic compliance with EU pharmaceutical regulations.
- Context Bridging Pharma: intelligent cross-system document search (LIS, SAP Pharma, eCRF) with over 98% accuracy, reducing technical information retrieval times by 45% and eliminating manual errors.
- Smart Folders GDP: automatic organization of batch documentation with real-time alerts on deadlines and deviations, ensuring complete traceability of batch records and compliance with EU serialization.
- Native Integration Hub: preconfigured and secure connectivity with major pharma enterprise systems (LIS, SAP Pharma, MES, Track & Trace, eCRF platforms), implementable in less than 30 days without interrupting production flows, and with documented ROI.
The distinctive Made in Italy methodological approach guarantees digital sovereignty over sensitive data, exclusive hosting on Aidia's physical servers in Italy, and dedicated expertise with highly specialized engineers, able to fully understand and support pharmacovigilance and serialization processes.
Use cases
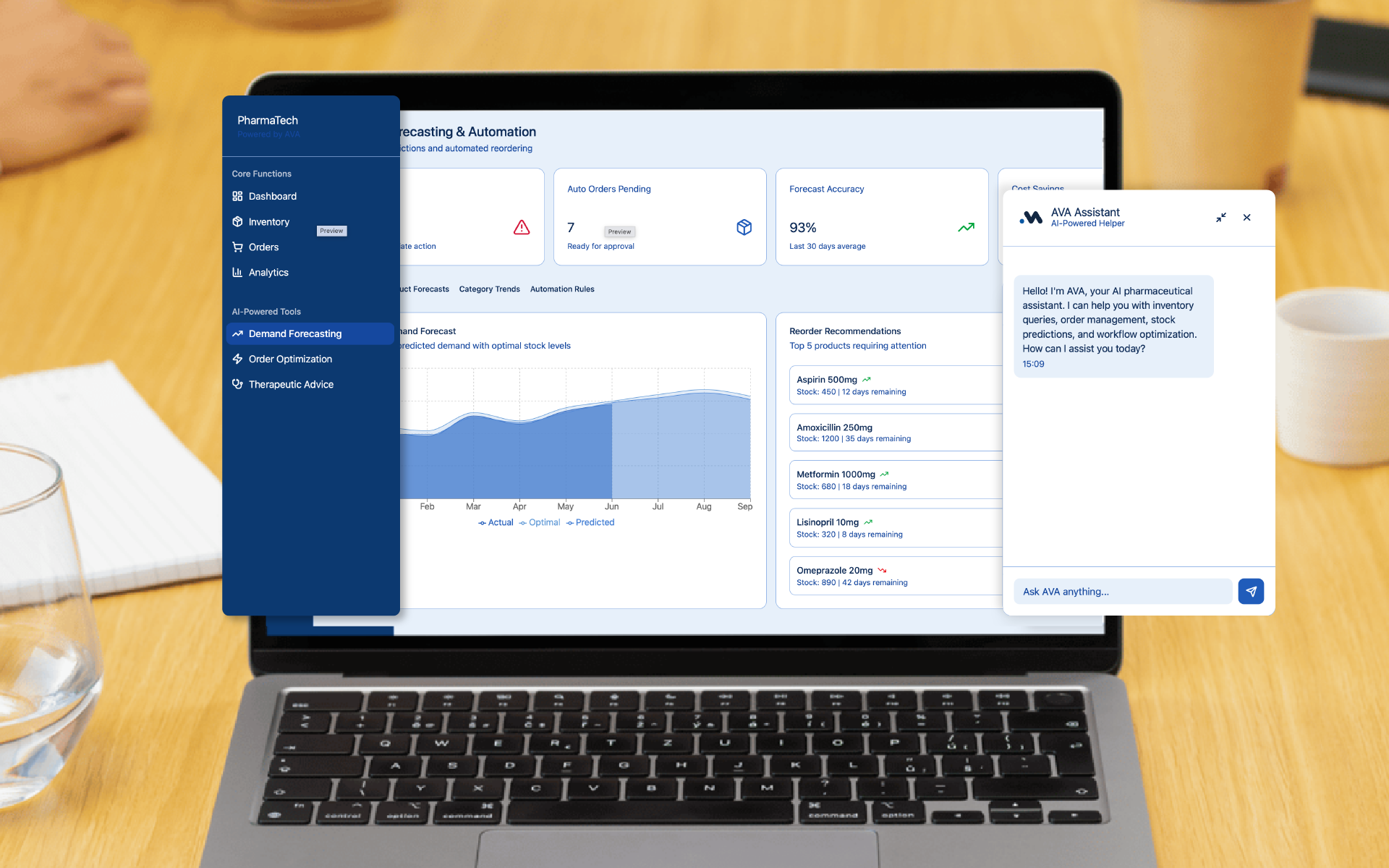
AVA for Regulatory Affairs Document Management in the Pharmaceutical Sector
Discover how we created an intelligent ecosystem for global regulatory compliance, integrating FDA, EMA, Health Canada, and PMDA into a single platform.

Document management through integrated artificial intelligence in pharmaceutical research
Discover how a pharmaceutical laboratory revolutionized knowledge management in R&D, thanks to AVAPharma.

AVA for Regulatory Affairs Document Management in the Pharmaceutical Sector
Discover how we created an intelligent ecosystem for global regulatory compliance, integrating FDA, EMA, Health Canada, and PMDA into a single platform.

Document management through integrated artificial intelligence in pharmaceutical research
Discover how a pharmaceutical laboratory revolutionized knowledge management in R&D, thanks to AVAPharma.
